CBD – So Many Questions, So Few Answers
Betty Chaffee/ August 28, 2020/ CBD, Dietary Supplements, Medication safety, Self management/ 4 comments
CBD, or Cannabidiol, is a popular newcomer to the healthcare marketplace. You've probably seen advertisements on the windows of retailers of all kinds, random text messages, pop-ups on websites. You can purchase it at a variety of local stores and from internet retailers. But how did CBD get SO popular, SO quickly? And what do we really know (and not know) about it? This will be the first in a series of articles about CBD. We'll look at what it's used for, what products are out there, and how to decide if it's right for you. In this first installment, we'll explore reasons for the widespread availability of CBD and consider how that may affect its safety and efficacy.
What is CBD?
CBD is one of many similar chemicals (cannabinoids) found in plants of the Cannabis species. It's related to THC (delta-9 tetrahydrocannabinol), the psychoactive chemical in marijuana that causes the "high". But CBD doesn't have psychoactive effects like THC. CBD and THC are produced in the same species of Cannabis, but in very different amounts depending on how the plants are cultivated.
Most THC is found in the flowers and leaves of the plant, so some plants are grown carefully and slowly to produce lots of flowers. This type of cannabis is known as "marijuana". On the other hand, CBD is present in many parts of the plant, parts that are useful for industrial purposes like making rope, clothing, paper, and more. For industrial purposes, plants are cultivated to grow quickly rather than produce a lot of THC. Cannabis grown in this way has a very low concentration of THC (<0.3%) and is known as "hemp". It became legal to grow industrial hemp in the United States almost two years ago, so now there's an abundant supply of CBD.
A Brief History of CBD
CBD was first identified in 1940. In 1970 Cannabis and all its chemicals (including THC and CBD) were made illegal in the US. That made it impossible to conduct scientific research on it. But many people believed all along that cannabinoids could improve the health of people with problems like sleep disorders, anxiety, epilepsy, and chronic pain. When hemp was made legal in 2018, it was a rush to the market for those who'd been waiting for years to sell CBD legally. And they did just that, resulting in sudden and wide availability of CBD products.
Regulation of CBD products
At about that same time, in 2018, the US Food and Drug Administration (FDA) approved a concentrated form of CBD for use as a prescription medication. It's called Epidiolex, and is used to treat patients with rare seizure disorders. But no other CBD formulation is FDA-approved. FDA did forbid the marketing of CBD as a dietary supplement or an additive to food products. Other than that the sale of CBD is unregulated at the federal level.
This is an unusual situation. Take the example of medicines for pain like ibuprofen (Advil) or naproxen (Alleve). These drugs were first approved by the FDA as prescription-only. After some years the FDA agreed that lower doses could be sold without prescriptions, and now we can purchase them over-the-counter. But they're still regulated by FDA, so there are standards that have to be met in testing, labeling, and so forth. On the other hand, CBD products were marketed directly to consumers before the FDA even approved the first prescription formulation. Now the FDA has to figure out the best way to ensure patients' health and safety. For now, non-prescription CBD is regulated only at the state level, and some states haven't weighed in on it yet.
Safety and purity of CBD - What's a Certificate of Analysis?
Lack of regulation causes some problems. We've talked about this in the past as it relates to dietary supplements. With dietary supplements, though, FDA does maintain some responsibility, it's just much looser than with medications. Conversely, non-prescription CBD currently isn't regulated at all by the FDA, so it can be produced without good manufacturing processes. Companies can market CBD without testing it for purity and potency, and they can put misleading information on the label. Don't get me wrong, many CBD manufacturers are meticulous about their product, and are honestly trying to provide a product that will improve the health of consumers. But there's no way to know for sure without going a little deeper. It's up to you, the consumer, to do your homework and make sure your CBD product contains the ingredients you're looking for AND is free from contaminants.
Reputable growers/manufacturers have their products routinely tested by an outside laboratory for potency and purity. In fact, you should be able to find a Certificate of Analysis (COA) for every batch (lot) of every product sold.
A sample COA is below. You can see by the heading of this COA that the information refers to one specific batch of product. The first page lists all the cannabinoids and the amount of each found in the product. This is where you can confirm that your product contains little or no THC, and find out whether it's pure CBD or contains other cannabinoids. Beneath that analysis you'll find details about possible microbial contamination, which lists each entry as a pass/fail.
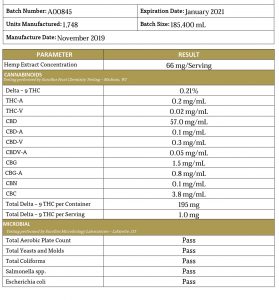
The second page of this COA gives a listing of possible contaminants with a pass/fail result on each. Cannabis is known for its ability to absorb many chemicals from the soil in which it grows, including heavy metals and pesticides. In order to be sure your product is safe, it must be free from these types of impurities.
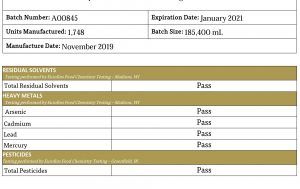
Different companies may use different formats, but the information should be the same. You want to know the active ingredients and if there's contamination, microbial or chemical.
If you've decided to try CBD, read the COA carefully to learn what cannabinoids the product contains and be sure it's free from contamination. No batch-specific COA available? Maybe best to find a different grower/manufacturer. COA results don't make sense? Ask your pharmacist about it or Contact Us!
We'll continue to talk about CBD on the BetterMyMeds blog. What kinds of products are out there, what ingredients to look for, the evidence showing that CBD improves health, and more. If you have a question or comment, remember that other readers might be thinking the same thing! Please post your comments and questions right here, or Contact Us directly at BetterMyMeds.



Great article on an interesting topic, Betty!
Along with the fact that most CBD products are not FDA-regulated (as you mentioned), there are WIDE variations regarding recommended doses of CBD for different uses (conditions). Additionally, as CBD is metabolized by liver enzymes, it’s important to know that using CBD with certain prescription medications could potentially result in drug-drug interactions.
Looking forward to reading the next post on this topic!
Pingback: CBD Products - What's Really in that Container? - BetterMyMeds
Pingback: CBD Oil - What are the Health Benefits? - BetterMyMeds
It was great meeting you at the Health Fair and the discussion about cbd. Feel free to contact me for more accurate information about my experience with it.